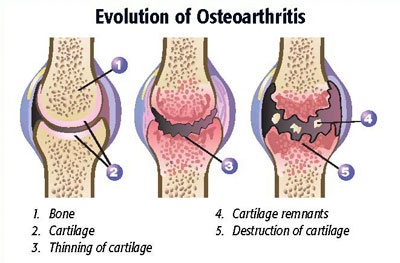
Medicine changes – information is added – our knowledge base grows. I wrote this article in 2014 – it’s time for an update, based on some new information.
Many, many people have asked me about Tildren®, the drug of the bisphosphonate class that was previously only available on import license from Europe. Now, the drug has been approved for use in horses in the US, along with another, similar drug, called Osphos®.
I wrote about Tildren® back in March, 2014. You can CLICK HERE to read the article. With the new drug approval, both Tildren® and Osphos® are probably going to be prescribed with increasing frequency. But there’s lots of stuff that I think you should know.
 SAFETY: The United States Food and Drug Administration has a site that is designed to give veterinarians safety information about the safety of the two drugs. You can see it if you CLICK HERE. The information shows that:
SAFETY: The United States Food and Drug Administration has a site that is designed to give veterinarians safety information about the safety of the two drugs. You can see it if you CLICK HERE. The information shows that:
- You shouldn’t use either drug while horses are on non-steroidal anti-inflammatory drugs (e.g., phenybutazone) because they increase the risk of kidney failure by a good bit.
- Horses receiving Tildren® colic with some frequency, and sometimes go off feed, or get a sore neck. To be more precise, ” However, this dosing regimen may be associated with a high incidence of colic signs. Treatment of colic signs with NSAIDs may adversely affect renal function. Long term effects on renal function are unknown.”
- Horses receiving Osphos® colic with some frequency, but, apparently, they rarely get sore necks (curiously, Osphos® is given in the muscle – Tildren® is given in the vein). To be more precise, “Clinical signs associated with administration of OSPHOS® included colic, agitation, and mild neurologic signs, such as tongue rolling and head shaking, that resolved within 5.5 hours following administration.”
EFFECTIVENESS: The FDA website doesn’t give you any information at all about how well the drugs actually work. For that, you have to go to the bottom of the page, for the Freedom of Information Act requests.
CLICK HERE for the FOI Act summary for Tildren®
- There were 181 horses evaluated in the Tildren® study (they started with over 800, but for various reasons, that’s what they ended up with.
- The majority of horses enrolled in the study had a history of navicular disease of less than 6 months duration
- Horses were examined about two months post treatment. Horses were deemed a treatment success if there was an improvement of 1 grade in lameness assessment.
And the results? Well, 48.39% of placebo-treated horses were deemed better after treatment, when looked at a couple of months later, and 63.87% of drug-treated horses were deemed better after two months.
 I KNOW WHAT YOU’RE THINKING. You’re probably thinking, “Well, 63.7% is better than 48.8%, so it might be worth a try.” And I understand that. Still, when we do studies, we analyze them to see if the number that look different actually are different. There’s a whole field devoted to that; it’s called statistics. And when they evaluated those numbers that I just cited, they found that they were barely significant, from a statistical point of view. Otherwise stated, there’s not much of a difference between treated and untreated horses when you looked beyond the raw numbers. Oh, and treated horses also had shoeing changes (the studies apparently didn’t attempt to see if horses got better from the shoeing changes, or from the drug, or both).
I KNOW WHAT YOU’RE THINKING. You’re probably thinking, “Well, 63.7% is better than 48.8%, so it might be worth a try.” And I understand that. Still, when we do studies, we analyze them to see if the number that look different actually are different. There’s a whole field devoted to that; it’s called statistics. And when they evaluated those numbers that I just cited, they found that they were barely significant, from a statistical point of view. Otherwise stated, there’s not much of a difference between treated and untreated horses when you looked beyond the raw numbers. Oh, and treated horses also had shoeing changes (the studies apparently didn’t attempt to see if horses got better from the shoeing changes, or from the drug, or both).
Next? CLICK HERE for the FOI Act summary for Osphos®
- There were 149 horses in the Osphos® study.
- Horses were deemed a treatment success if there was an improvement of 1 grade in lameness assessment at 56 days after treatment.
- At 180 days, some of the horses that were deemed better at 56 days weren’t better anymore.
And the results? Well, if you look just on day 56, the treatment seemed to be wildly successful. Incredibly successful, actually. Over 70% of the horses treated with Osphos® got better, compared to only 3% getting the placebo. In fact, if you take the numbers at face value, the treatment is probably more incredibly successful than anything else that’s ever been described for treating navicular disease.
Which I wonder about.
MY CONCERNS
 I wouldn’t be me, I guess, if I didn’t wonder about these studies a good bit. So, as briefly as I can, let me tell you about some things that bug me about these study summaries.
I wouldn’t be me, I guess, if I didn’t wonder about these studies a good bit. So, as briefly as I can, let me tell you about some things that bug me about these study summaries.
- I find the ages of the horses studied to be a bit weird in both of the studies. The horses in the two studies ranged from 4 – 22 years of age. To me, that’s odd, since navicular disease is generally a disease of 7 – 11 year old horses, but OK.
- The drug dosing used for Tildren in the US is different than that which has been approved in Europe. Same drug – different dosing schedule. I don’t know why.
- I’m not convinced that all of the horses in the study even HAD navicular disease, particularly in the Osphos® study, where the inclusion criteria was a lot looser (they had MRIs in the Tildren® study, but not in the Osphos® study), but also in the Tildren® study, where navicular disease was diagnosed in at least some horses based on edema (swelling) of the navicular bone; it’s not the same thing. Horses in the Osphos® study were diagnosed with navicular disease based on response to a nerve block, as well as X-ray changes, some of which aren’t necessarily diagnostic for navicular disease (e.g., “elevated number of lucent zones”).
- If either drug is going to work, it’s apparently going to take a while.
- “Work” doesn’t necessarily mean that your horse is going to be a lot less lame. The lameness grading scale is super subjective – it’s hard to really say what a change of 1 grade really means in many cases. If your horse goes from a 4 to a 3, or a 3 to a 2, he’s still pretty lame.
- As far as I can tell, no one was blinded in the studies. That means that observers knew who go the drug, and who didn’t. If that’s true, it has a lot of implications for the study, that is, there’s a lot of potential for biased results. NOTE: 8.7.2014: Based on a comment from a rep from the company that makes Tildren®, the observers were blinded.
- In the Tildren® treated horses, nearly 50% responded to placebo. In the Osphos® study, only 3% responded to placebo. Why the big difference, if they had the same condition at the start? Either Osphos® is a miracle drug, or there’s something a bit screwy going on.
- I worry about picking one time point to evaluate something like lameness. If you’ve had a lame horse, you’ll probably find that he’s better on some days than others. I would have liked to see multiple evaluations. And, multiple evaluators, who didn’t know which horses got which drug. But that didn’t happen.
So, basically, based on all of this new information, I’m pretty unimpressed. It’s a little frustrating, looking at the summaries of the studies, because there’s not a lot of detail. Mostly, to me, all of this new information means that the drugs are going to be easier to get.
Bottom line? The drug companies got the drugs approved. It looks to me like they got approved on some fairly thin criteria. They got it approved to treat one thing, navicular disease. Legally, however, veterinarians can use it at their discretion to treat just about anything that they want. You’ll likely find that many of them will.
 My guess is that, fairly quickly, Osphos® will become the more popular drug. I think that’s because Osphos® is easier to give than Tildren®. Osphos® is given in the muscle and. Tildren® is given by slow intravenous infusion. And it’s cheaper, too. I think that the combination of easier to give and cheaper means that it will probably be used more in the long run.
My guess is that, fairly quickly, Osphos® will become the more popular drug. I think that’s because Osphos® is easier to give than Tildren®. Osphos® is given in the muscle and. Tildren® is given by slow intravenous infusion. And it’s cheaper, too. I think that the combination of easier to give and cheaper means that it will probably be used more in the long run.
Hopefully, as the FDA website assures us, these drugs will be monitored after-market. What we mostly have now is some studies sponsored by the companies selling the product. Based on those studies, the drug got approved for sale in the US. At this point, call me unimpressed.
UPDATE – 3.22.2018
And you can still call me unimpressed. Since I wrote this article, there hasn’t been that much new information. But there have been three bits of information that I consider pretty important.
The first one is just released. In a 60 day study of 19 Thoroughbred horses, performed at Louisiana State University, researchers concluded, “Tiludronate and clodronate do not appear to significantly impact bone tissue on a structural or cellular level using standard dose and administration schedules.” Tiludronate is Tildren® and clondronate is Osphos®. Otherwise stated, using standard doses, researchers couldn’t find that either drug did anything to bone. That’s a big deal, really, since the point of the drugs is to do something to bone.
 The next one are comments from Dr. Larry Bramlage, former president of the American Association of Equine Practitioners, and one of the more important and influential surgeons who has ever put on a cap and gown for equine surgery. These comments are published in the Paulick Report, which is an influential reporting service in the Thoroughbred Racing Industry. In a presentation that Dr. Bramlage recently gave, he said, ““I wish we’d never seen these drugs.” He feels that they are responsible for delayed bone healing, and he’s concerned (as I am) about the indiscriminate use of these drugs. I’ve heard of people using them for “bone maintenance” – that’s just stupid. Anyway, if Dr. Bramlage doesn’t like them, I pay attention.
The next one are comments from Dr. Larry Bramlage, former president of the American Association of Equine Practitioners, and one of the more important and influential surgeons who has ever put on a cap and gown for equine surgery. These comments are published in the Paulick Report, which is an influential reporting service in the Thoroughbred Racing Industry. In a presentation that Dr. Bramlage recently gave, he said, ““I wish we’d never seen these drugs.” He feels that they are responsible for delayed bone healing, and he’s concerned (as I am) about the indiscriminate use of these drugs. I’ve heard of people using them for “bone maintenance” – that’s just stupid. Anyway, if Dr. Bramlage doesn’t like them, I pay attention.
Last is a study from 2016, where researchers studied two ways to give Tildren®, in lame horses. There were 12 horses in the study, all of which were diagnosed with navicular disease. Some horses got the medication intravenously – others got it by a technique called regional limb perfusion, where you put a tourniquet on the horse’s leg and then run the drug into the leg below the tourniquet. The idea is that the drug sort of soaks into the area since the blood isn’t moving much. It’s not a use that is recommended by the manufacturer, but that’s another story. Anyway, the results in these 12 horses were that the horses were still lame after treatment. Better, perhaps, but still lame.
 So, honestly, I don’t get it. I mean, I do get it – everyone wants to try to help horses, and we need new and better ways to do it. But I don’t get the enthusiasm for these drugs, I don’t get the off-label use for conditions where it “might” help, and I don’t get why people haven’t been more careful with them. Honestly, I think the reason why there haven’t been more problems is that they are old drugs that are being used at a dose that’s not likely to have much of an effect (see the LSU study).
So, honestly, I don’t get it. I mean, I do get it – everyone wants to try to help horses, and we need new and better ways to do it. But I don’t get the enthusiasm for these drugs, I don’t get the off-label use for conditions where it “might” help, and I don’t get why people haven’t been more careful with them. Honestly, I think the reason why there haven’t been more problems is that they are old drugs that are being used at a dose that’s not likely to have much of an effect (see the LSU study).
Like I said, I’m not a fan. If your horse has been helped, I think that’s great. Just be careful, please. Your horse is counting on you.